Abstract
Background:
The genetic hallmark of BL is MYC gene rearrangement. However, due to histopathological, immunohistochemical and cytogenetic similarities, differentiation from Diffuse Large B-Cell Lymphoma (DLBCL) remains challenging in some cases. Although being suspected to negatively influence outcomes, detection of high expression of BCL2 and MYC (double expressor) as well as rearrangements of MYC and BCL2 and/or BCL6 (double/triple hit) did not lead to confirmation of different therapeutic approaches in DLBCL yet. The recent update of the World Health Organization (WHO) classification of lymphoid neoplasms has thus established a category termed High-Grade B-Cell Lymphoma (HGBL), defined by a distinct morphology and/or presence of double/triple hit status. While BL displays favorable outcomes in clinical routine upon treatment with intensive immunochemotherapeutic regimens, clear evidence regarding the optimal therapeutic approach for HGBL remains limited and the diagnostic discrimination of BL and HGBL can be challenging, too. Moreover, data on the frequency of revisions of the diagnosis of BL to DLBCL or HGBL and its impact on treatment and outcome are sparse.
Methods:
All consecutive patients managed with histopathological suspected diagnosis of BL upon preliminary assessment at the reporting institution between December 2010 and July 2020 were identified. Final diagnosis of the respective aggressive B-Cell lymphoma was grouped according to the most recent WHO classification and divided into four subgroups: a) BL, b) revision from DLBCL to BL, c) DLBCL, d) HGBL. General patient characteristics as well as response to treatment in Computed Tomography (CT) or Positron Emission Tomography (PET) (evaluation by individual investigator), information regarding survival supported by the register of deaths in North-Rhine Westphalia and the date of last follow-up were collected from individual patient files.
Results:
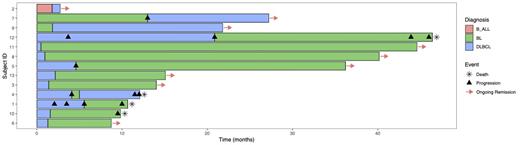
Overall, we identified n=66 patients with suspected diagnosis of BL within the preliminary assessment. Final histopathological results confirmed n=31 patients as BL (Group A), while n=23 patients were described as showing features of both BL and DLBCL (Uncertain & Grey-zone, Group D). Additionally, in n=12 patients the final diagnosis was revised either from DLBCL to BL (n=9, Group B) or B-ALL/BL to DLBCL (n=3, Group C). Regarding the reference histopathological reviews (RHR) and the latest version of the WHO classification of lymphoid neoplasms, all patients from Group C and Group D were finally classified as either DLBCL (n=12) or HGBL (n=14). In total, n=13 patients had revision of the final diagnosis, either upon receipt of RHR (n=8) or repeated biopsy upon progressive disease (PD) (n=5). When comparing patients with revised diagnosis due to PD or RHR, a median of six (range 4-8) and two (range 1-2) cycles of systemic therapy were applied prior to revision, respectively. Regarding outcomes, 2/5 patients (40%) with revised diagnosis upon PD and 7/8 patients (87,5%) with revised diagnosis upon RHR were alive at the time of last follow-up (median follow-up 15,07 months), respectively (Fig 1). Patients suffering from BL (Group A) displayed a significantly increased progression-free survival (PFS) compared to all other groups (p=0.034). In contrast, patients whose diagnosis was revised from DLBCL to BL after initiation of treatment with DLBCL protocols had an inferior PFS than patients initially diagnosed with BL (p=0.045) that was comparable to patients with the final diagnosis of DLBCL or HGBL. Two-year overall survival was generally favorable in all groups, ranging from almost 60% (HGBL) to 86,2% (BL) without showing statistically significant differences (p=0.2).
Conclusion
In conclusion, timely and precise histopathological diagnostic procedures seem to play a critical role for optimizing treatment strategies of BL, HGBL and high-risk DLBCL upfront. Since administration of BL treatment protocols seems feasible in clinical routine and may result in favorable results not only in BL but as well in HGBL, these results might encourage its upfront usage in doubtful situations regarding histopathological results. However, with only a small number of patients being evaluable in the present analysis, controlled clinical trials are necessary to confirm these trends.
Heger: Novartis: Research Funding; Gilead: Other: Travel funding. Gödel: Gilead: Other: Personal fees / travel support; Novartis: Other: Travel support. Balke-Want: Novartis: Research Funding. Simon: Gilead: Other: Travel support. Bröckelmann: BMS: Honoraria, Research Funding; Takeda: Honoraria, Research Funding; MSD: Research Funding; BeiGene: Research Funding. Von Tresckow: Amgen: Consultancy, Honoraria; AstraZeneca: Honoraria, Other: Congress and travel support; AbbVie: Other: Congress and travel support; BMS-Celgene: Consultancy, Honoraria, Other: Congress and travel support; Kite-Gilead: Consultancy, Honoraria; MSD: Consultancy, Honoraria, Other: Congress and travel support, Research Funding; Novartis: Consultancy, Honoraria, Other: Congress and travel support, Research Funding; Pentixafarm: Consultancy, Honoraria; Pfizer: Consultancy, Honoraria; Roche: Consultancy, Honoraria; Takeda: Consultancy, Honoraria, Other, Research Funding.